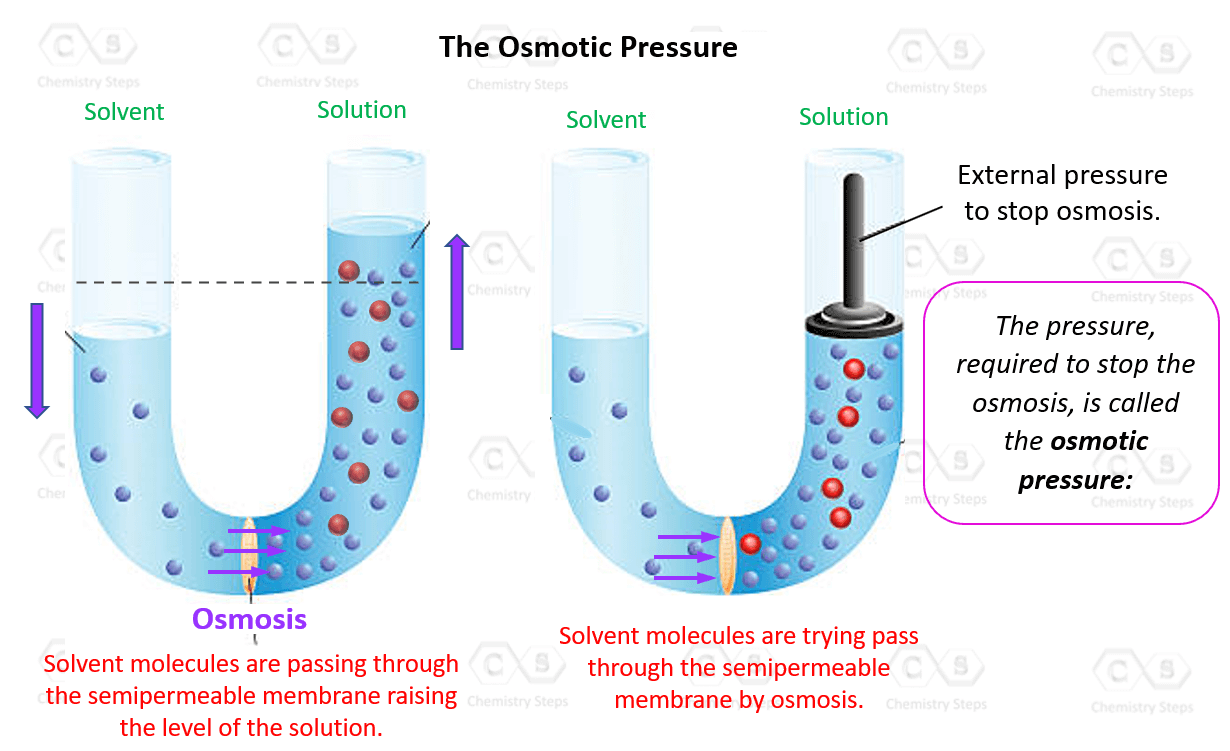
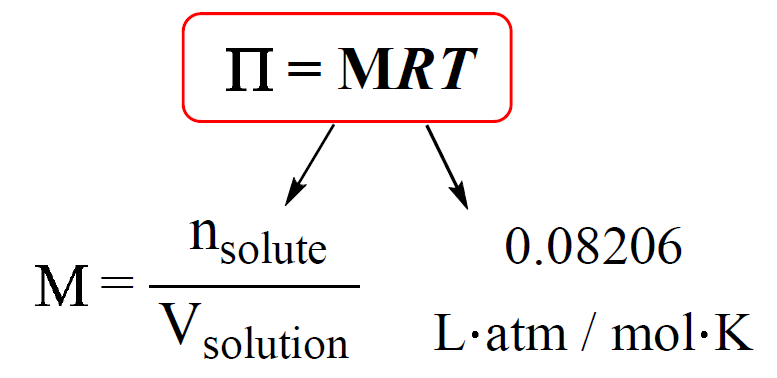
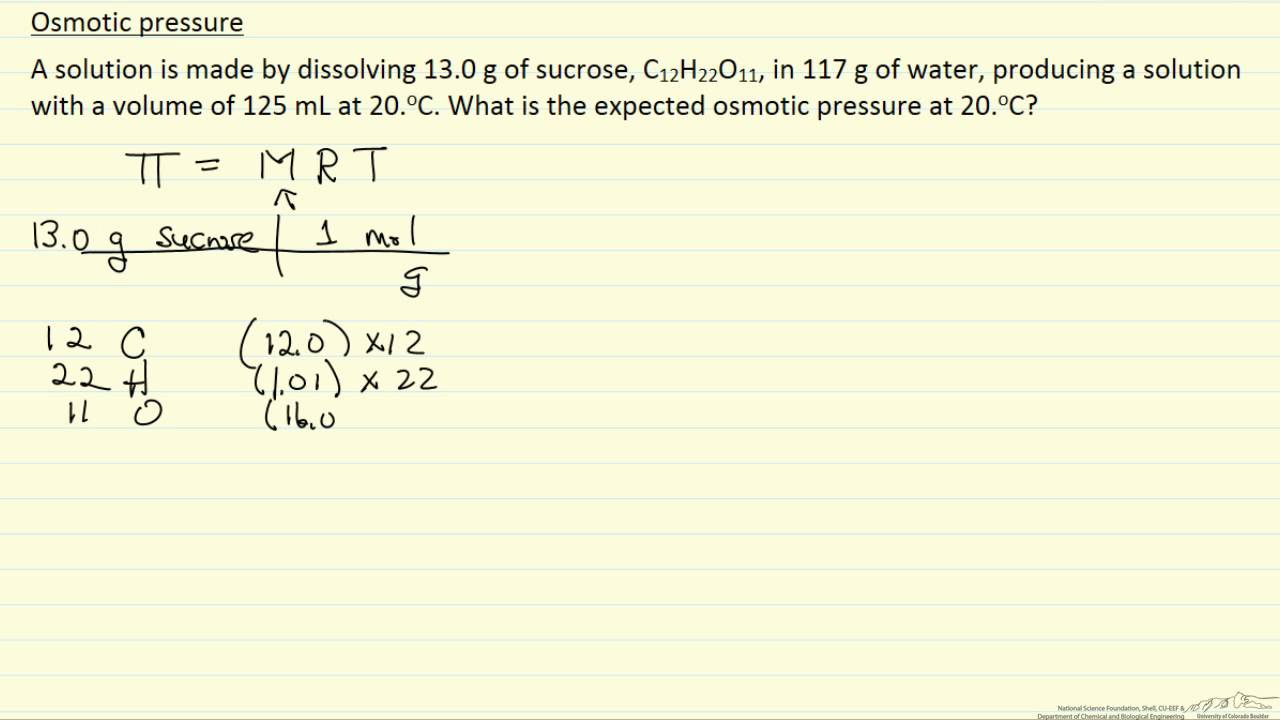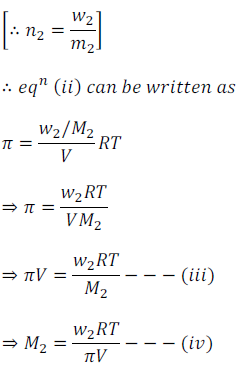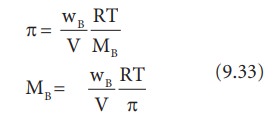OneClass: calculate the osmotic pressure of a 0.100 M aqueous solution of NaCl at 25°C and the molar...
Calculate the osmotic pressure in pascals exerted by a solution prepared by dissolving 1.0 g - Sarthaks eConnect | Largest Online Education Community
![Calculate the osmotic pressure exerted by a solution prepared by dissolving 1.5 g of a polymer of molar mass 185000 in 500 ml of water at 37^o C. [R = 0.0821 L.atm.K^-1.mol^-1 ] Calculate the osmotic pressure exerted by a solution prepared by dissolving 1.5 g of a polymer of molar mass 185000 in 500 ml of water at 37^o C. [R = 0.0821 L.atm.K^-1.mol^-1 ]](https://i.ytimg.com/vi/PtV0tDX6lCI/maxresdefault.jpg)
Calculate the osmotic pressure exerted by a solution prepared by dissolving 1.5 g of a polymer of molar mass 185000 in 500 ml of water at 37^o C. [R = 0.0821 L.atm.K^-1.mol^-1 ]
Calculate the osmotic pressure of 5% solution of urea at 273K. - Sarthaks eConnect | Largest Online Education Community
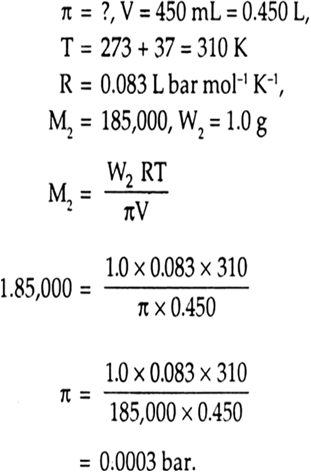
Calculate the osmotic pressure in pascals exerted by a solution prepared by dissolving 1.0 g of polymer of molar mass 185,000 in 450 mL at 370C. from Chemistry Solutions Class 12 CBSE

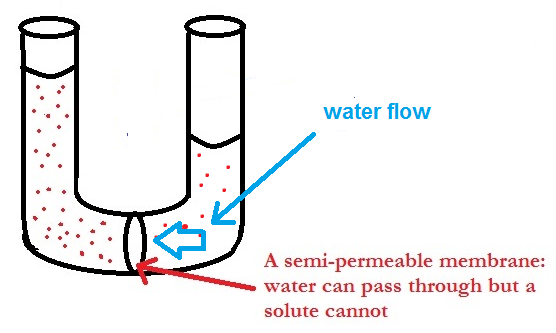
Twitter 上的DocMD #LENI2022 ✊🏼🇵🇭 💗:"25.Osmotic Pressure-pressure needed to prevent osmosis, which is the spontaneous net movement of solvent molecules through a semi-permeable membrane in to a region of higher solute concentrations.